I think most people went through (and are still going through) a reflective period during this pandemic and times of isolation. We stayed home longer than ever before, deprived of day-to-day routines and activities, and were left to ponder our own value and individual purpose. Covid-19 is not an alien invasion, nor some other existential crisis, but a biological hazard that we will come to better understand how it infects, how to prevent its spreading, and potentially how to eradicate it. It is good to remember that these times are unprecedented and it is okay to have feelings of uncertainty. We are all in this together (not to quote Red Green…https://en.wikipedia.org/wiki/The_Red_Green_Show) and the best thing is to find ways to stay comfortable, relaxed and occupied, either with some work, new/old hobbies or anything you enjoy.
During confinement, I was in a fortunate position to be able to continue my work from home. I sifted through data that I had accumulated over the past two years, analyzing data and working up results. Through video conferencing it was possible to stay connected with others in the research group and to feel somewhat motivated and focused on work-related tasks. I will admit that working from home had some perks in the first few weeks, but after a couple of months it became a chore, trying to find ways to coax myself into work-mode. My level of motivation dipped and the line separating work from leisure became blurred. Alas, the conditions for working were far from normal so the expected amount of productivity should be scaled accordingly for anyone in the same position. Luckily, I was able to return to the lab in June and have been going steadily since. With the current second wave of cases and hospitalizations, who knows how long that will last.
Due to SARS-CoV-2, my last beamtimes scheduled for June and July were postponed until the late summer and early 2021. So, while my own measurements are on pause, I decided to write about a couple of beamtimes that I participated in as a colleague and collaborator over the last couple of years. One opportunity attached to research is the potential for collaboration and for sharing ideas and insights. Sometimes applying your knowledge and skills to a new set of problems outside of your own daily research tasks can lead to new perspectives and findings. I was grateful to be involved in two projects on the formation of synthetic magnetite (Fe3O4) nanoparticles, which used X-ray absorption fine structure spectroscopy (XAFS, how this technique works was previously described in the “Advanced Photon Source: …” post) to help understand nanoparticle growth mechanisms (how individual Fe ions come together to form small iron oxide clusters, and proceed to final larger Fe3O4 nanoparticles). These nanoparticles are essentially the same material produced by magnetotactic bacteria (the famous critters I have been talking about in previous posts), but they are instead formed purely by chemical means with no biology involved. Primarily, the synthetic magnetite nanoparticles examined in these works were of interest to understand fundamental processes governing formation and structure, but the materials themselves have application as catalytic materials, magnetic data storage devices and for biomedical application (hyperthermia treatment of cancerous tissue and magnetically-guided drug delivery, for example).
The first project and XAFS beamtime I will discuss was led by Dr. Lucas Kuhrts, a freshly minted PhD from the Max-Planck Institute for Colloids and Interfaces (MPICI, where my own post doc project began in 2018). Lucas had been studying the formation of magnetite nanoparticles with the addition of polymers (long, linear chains of repeated chemical groups), which changes the way smaller iron oxide clusters form larger nanoparticles. When I had first met Lucas, he had already performed a thorough investigation of the polymer-magnetite system with small angle X-ray scattering (SAXS), a technique that measures how incident X-ray light is scattered by particles in the sample, giving information on the size, density, and shape of those particles. In order to complete his formation mechanism, I suggested to assist in performing XAFS measurements to learn more about the local environment of iron atoms and the particular phase of iron oxide that may exist at various time-points in the synthesis. Upon agreement of applying XAFS to help solve his problem, we wrote a beamtime proposal and were awarded a few days at the Diamond Light Source in the UK (where I had already performed X-ray microscopy measurements, as shown in previous posts).
Fast-forward a few months after our awarded beamtime notice and here we were at the I-20 Scanning beamline of Diamond Light Source. Along with Lucas and I, was Oliver Späker, a PhD researcher also from MPICI. The important questions we wanted to answer regarding the formation of polymer-magnetite nanoparticles took place on a time-scale of minutes, where the crucial formation steps occur in the first minute to several tens of minutes after iron precursors, polymer, and base are added together for the chemical reaction and formation of magnetite, which proceeds as follows:
2FeCl3(aq) + FeCl2(aq) + 5NaOH(aq) >>> Fe3O4(s) + 5NaCl(aq)
You will see two different iron chloride salts for the reactants, which are the valence states of 2+ and 3+ needed to form the mixed valence that is magnetite (Fe(II)Fe(III)2O4). Considering the rate at which we could collect a single spectrum at I-20 Scanning for our dilute samples (low concentrations of Fe, with ~10-15 minutes per spectrum and several would need to be collected to improve signal to noise) and the several early time points we wanted to investigate, an in situ measurement was not feasible (this is where the measurements would be conducted on a sample environment where the chemistry is happening in real-time). This would mean that by the time we started and finished the collection of just one spectrum, the formation of magnetite nanoparticles would have been nearly complete. Therefore, we took an ex situ approach (the samples to be measured were taken from the natural sample conditions at various time-points and then preserved for later measurement). Here, samples were extracted from the chemical reaction at the time points of interest and were immediately frozen on a sample holder using liquid nitrogen.

Side profile of Lucas Kuhrts in the chemical laboratory preparing a series of polymer-stabilized magnetite nanoparticles 
Side profile of Oliver Spaeker in the control room keeping the team organized with experimental conditions left to measure

Filling the sample chamber with liquid nitrogen to keep the extracted samples frozen and also to improve the quality of the collected data. 
An example of how mounted samples were prepared for measurement. This was a reference powder of hematite recently removed from the chilly liquid nitrogen sample chamber.
The truly heroic aspect of this beamtime was that Lucas had brought his experimental apparatus with him from Berlin to Oxfordshire. Since we were not exactly sure which time points would be of most interest to capture the formation mechanism with XAFS (somewhere between 1-20 minutes…), Lucas would synthesize a series of samples, we would measure them, analyze the data to understand how iron oxide particles were forming and growing, and then redo it over again until we had samples at various time-points that gave us an overall picture of the chemical and physical process. It was a lot of work and determination on Lucas’s part, where Oliver and I tried to keep a broader view of the experiments and measurements to make sure we would have enough data by the end of the beamtime to complete Lucas’s study. In the end, we came away with information we had hoped to gain. We were able to capture how the added polymer affects the formation of magnetite nanoparticles: by delaying the rapid formation of magnetite phase by a precursor iron oxide phase known as ferrihydrite that interacts with the added polymer. We were so pleased with ourselves that we brought the news and the experimental apparatus to the Queen in London…

Lucas and the titration apparatus used to produce magnetite nanoparticles in front of Buckingham Palace
The second collaborative beamtime was also conducted at Diamond Light Source, at the sister beamline of I-20 Scanning, named I-20 Energy Dispersive EXAFS (I-20 EDE). The leader of this beamtime was Dr. Jens Baumgartner, a postdoctoral researcher at the time from MPICI. Joining him were myself and Lucas. Jens had been studying the formation of magnetite for several years since his PhD and has become well known in the field of biomineralization, examining magnetite formation from both biological and synthetic perspectives. One of the long-withstanding questions surrounding magnetite formation, and other many other minerals for that matter, is the exact nature of precursors that first form (nucleation) and directed the formation of the final nanoparticle product (growth). Capturing this chemical and physical information of nanoparticle growth at very early stages would provide an intimate understanding on how to better control inorganic nanocrystal growth, which would open up new approaches to design advanced nanomaterials. Again, very fundamental research objectives, but the principle is to enable more synthetic control over nanoparticle formation, which would have broad implications for future nanotechnology.
To capture information on the fast chemical and physical formation of magnetite we really needed to measure what is occurring on the time scale of seconds. More like milliseconds, actually… And for this reason Jens had chosen the I-20 EDE beamline to conduct this XAFS experiment. The EDE technique specializes in collecting an entire XAFS spectrum in a matter of milliseconds with even microsecond time scales feasible with today’s detector technology (whereas in the above beamtime, it took about 10 minutes for one spectrum). Generally speaking, we were able to collect an entire spectrum 60000 times faster than with the previous XAFS technique! Of course, there are some disadvantages with using such a fast technique. Mainly, the concentration of you material or element of interest has to be at higher concentrations. The concept of the energy-dispersive measurement is quite interesting. The following image shows the principle behind the measurement. The general idea is that one spectrum can be collected instantaneously, in a matter of milliseconds, which is highly advantageous for capturing fast chemical formation processes.
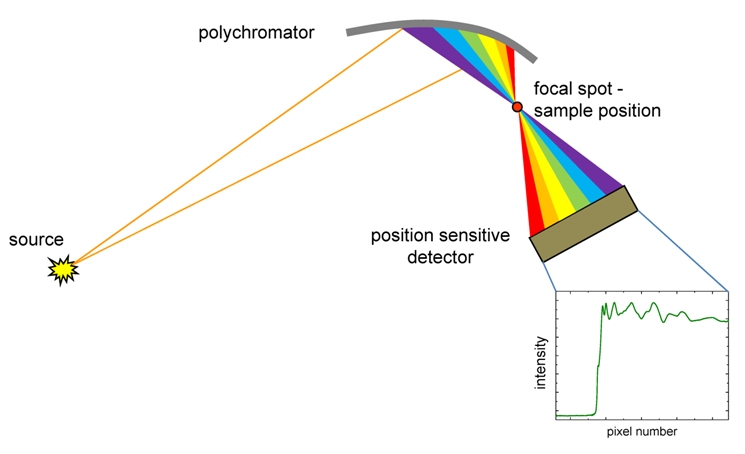
Image source: https://www.diamond.ac.uk/Instruments/Spectroscopy/Techniques/EDE.html
So with a fast method of collecting data, how to trigger the chemical reaction in a controlled and precise manner? For this a stopped-flow device was used, which can be seen in the images below. There, several plastic syringes can be seen installed into a stainless steel block and are controlled by a pressurized pump. On top and in the middle of this block is where solutions from the syringes are injected and subsequently mixed, contained inside a thin X-ray transparent tube (i.e., a capillary of Kapton polymer). As the solutions come in contact with one another in this tube, the formation of magnetite proceeds. It is also the exact position where the dispersed X-ray beam converges and then diverges toward the detector. The stopped-flow device is specially designed for the study of fast chemical processes because it can deliver highly accurate volumes of each solution into the measurement region and with coordinated precision to ensure the reaction will proceed in the same manner each repetition. The triggering mechanism can be controlled remotely, as we did this when we were safely outside of the experimental beamline hutch and in the control room. This way, we could trigger the reaction and start the XAFS measurement simultaneously.

Lucas and Jens running first tests on the stopped-flow device to ensure each solution is being triggered correctly. 
Lucas making adjustments to the stopped-flow device before exiting the hutch and starting measurements.
This beamtime was technically challenging. Also, an exhausting for the team since we had to enter the experimental hutch to recharge the solutions and make any minor adjustments to the stopped-flow device. One of the first things needed to be done was to determine the right concentration of Fe to use in the solutions. If the concentration was high, there was a good XAFS signal, but the formation of magnetite occurred too fast to capture. If the concentration was low, the X-ray absorption on the sample would be too weak, resulting in XAFS data that was too noisy to work with. After finding the right concentration, more problems would arise and it was a struggle to keep things going enough to collect meaningful data to complete a study. But thanks to a lot of coffee and several days of beamtime, things came together and we left with a lot of data… A LOT. I do not have the definitive numbers in front of me but we were collecting about 50000 spectra every 5-10 minutes and we had about 7 days of beamtime. Needless to calculate, we left with millions of spectra on the formation of magnetite! There are definitely some exciting things to come from the analysis of this data (from both beamtimes).
I look forward to sharing the next synchrotron trips and adventures with you, but I am not quite sure when that will be exactly. I had conducted a recent beamtime remotely from home, which was a great experience and could be the temporary future of synchrotron measurements… Anyway, we will see and I will update again soon with other synchrotron trips and recent events that I have yet to share. Until then, take care and wash your hands.





































